Atoms are most stable if they have a filled valence shell. 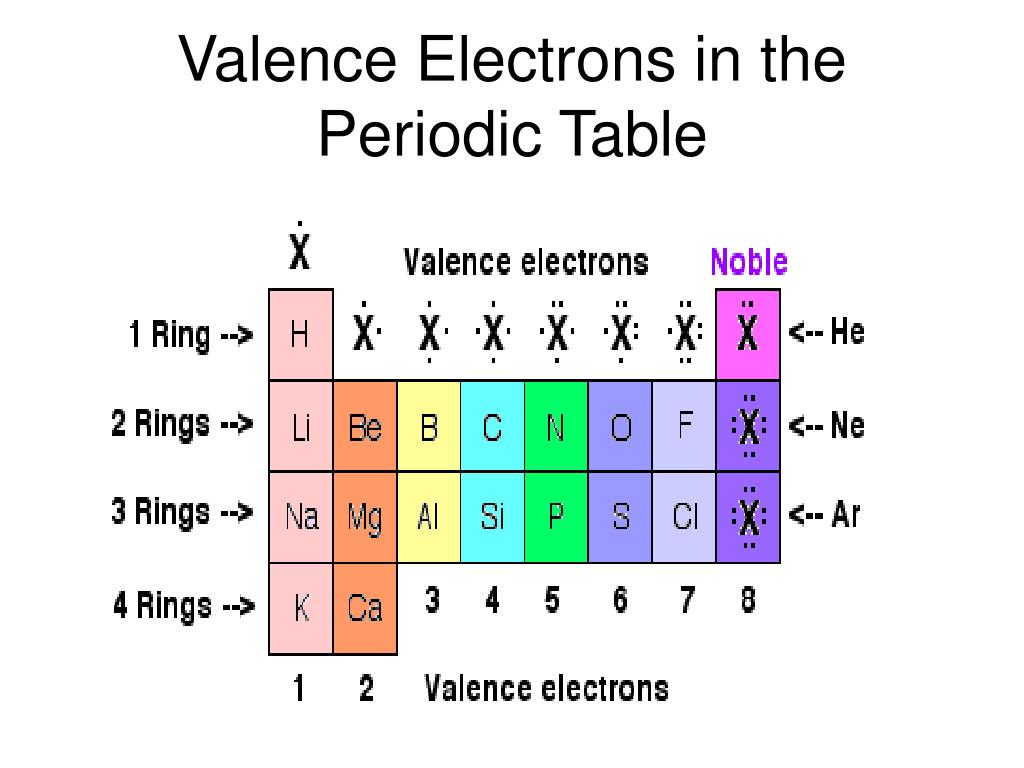
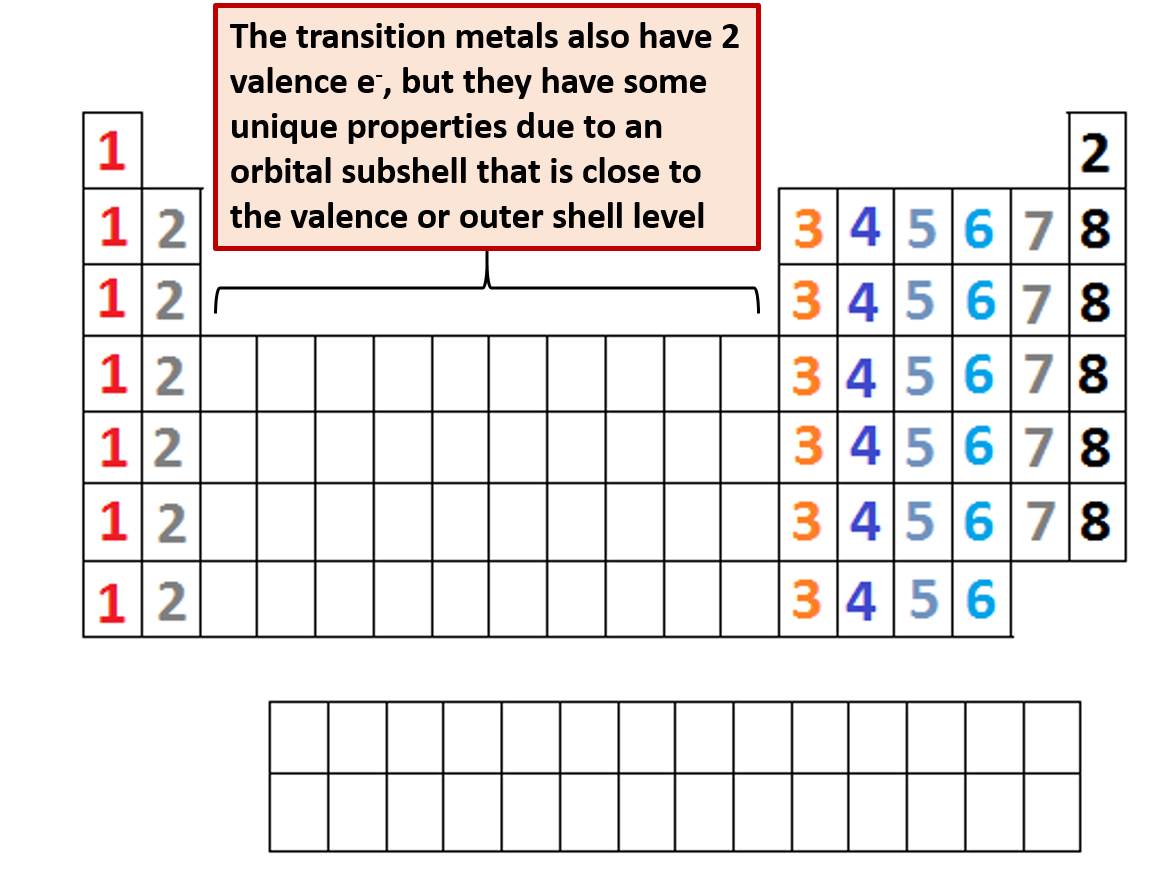
The number of valence electrons of an atom can be obtained from the periodic table because it is equal to the group number of the atom. It is said to occupy orbitals in an atom. Write the valence electron configuration of each element by first indicating the filled inner shells using the symbol for the nearest preceding noble gas and then listing the principal quantum number of its valence shell, its valence orbitals, and the number of valence electrons in each orbital as superscripts.Ī The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons. Electrons are involved in the chemical bonding and reactions of the atom.Locate the nearest noble gas preceding each element and identify the principal quantum number of the valence shell of each element. Identify the block in the periodic table to which the group 2 elements belong.Use the periodic table to predict the valence electron configuration of all the elements of group 2 (beryllium, magnesium, calcium, strontium, barium, and radium).Īsked for: valence electron configurations For elements after No, the electron configurations are tentative. In groups 112, the group number matches the number of valence electrons in groups 1318, the units digit of the group number matches the number of valence. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. \): Electron Configurations of the Elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
